 The teacher can follow this activity with other elements, too. ***Hint: To find the number of neutrons, students must subtract the atomic number from the atomic mass (9amu – 4protons = 5neutrons) What is this element? What are the atomic number, atomic mass, and element symbol? How many neutrons and electrons should there be? The teacher can call on more volunteers to pick up neutron and electron signs, and the volunteers must form the Beryllium atom. The unit of measurement is the ATOMIC MASS UNIT, or AMU.Īctivity 1: The teacher calls on 4 volunteers to pick up 4 proton signs. You can find it by adding the protons and neutrons together. The ATOMIC WEIGHT/ ATOMIC MASS is the average mass of an element. The ELEMENT SYMBOL is the element’s shorthand or abbreviation The ATOMIC NUMBER is the number of protons there are in the atom. 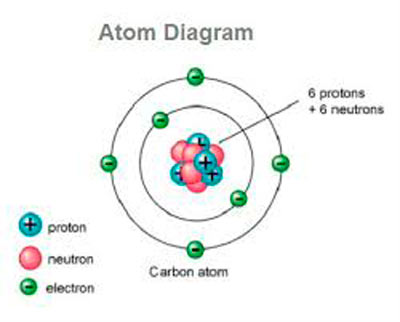
How many protons are in our atom? The volunteers formed the helium atom, which normally has 2 protons, 2 neutrons, and 2 electrons.ĭemonstration: The teacher shows the students how to use the periodic table. 
We can tell what element the atom is by counting the number of protons in the nucleus. In general, there are the same number of protons and electrons in an atom. The electrons orbit around the nucleus in electron shells. The students explain why they know which volunteer is which particle.Įxplanation: The protons and neutrons make up the nucleus, which is the core of the atom. Practice: The teacher calls on students to point to which volunteers are the proton, neutron, and electron particles. ***Helpful visual aid: Bohr’s Model of the atom They make up everything around you–including air! There are three basic parts of an atom: the PROTON, which has a positive charge the NEUTRON, which as no charge and is neutral and the ELECTRON, which has a negative charge. ***Students will be forming a helium atomĮxplanation: ATOMS are the basic building blocks of matter. The “protons” and “neutrons” stand together, while the “electrons” “orbit” around them. 
Helpful visual aid: Bohr’s Model of the atom Assignment:ĭemonstration: The teacher calls on 6 volunteers to the front of the classroom and hands a “proton” to 2 students, a “neutron” to 2 students, and 2 “electrons” to the last student. Periodic tables for students to reference later on in the activity. Three sets of signs: red with a plus (protons), yellow (neutron), and blue with a minus (electron). In the advanced version, students learn about isotopes and ions. They will also learn how to calculate the atomic mass and find the number of protons, neutrons, and electrons. In the basic version, students learn about the atom, its structure, the particles. This activity compliments the teacher’s lesson on the atom. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
